| Content Pages | ||
|
Social Complexity Social Inference Symbolic Inference |
||
|
|
||
| People | ||
|
Alan B. Bond Alan C. Kamil Russell P. Balda Jeffrey R. Stevens |
||
|
|
||
| Docs & Links | ||
|
Books Publications Links |
||
|
|
||
 |
||
Social Inference
Pinyon Jay Flock, © Steve Summers Living in stable social groups requires a range of cognitive abilities, including recognizing group members, tracking their social status, and inferring relationships among them (Kummer et al., 1997; de Waal & Tyack, 2003). Because the number of possible pairs of individuals ("dyads") increases rapidly with group size, members of large social groups cannot rely only on their own interactions with other individuals to make relationship judgments. Transitive reasoning should, therefore, be particularly important for social species (Seyfarth & Cheney, 2003), but its use in making social judgments had never been demonstrated in animals. Our experiment (Paz-y-Miño, Bond, Kamil & Balda 2004) showed that highly social pinyon jays (Gymnorhinus cyanocephalus) can draw sophisticated inferences about their own dominance status relative to that of strangers simply by observing the stranger's interactions with other, known individuals. These results provide the first clear demonstration that animals use transitive inference in social settings.
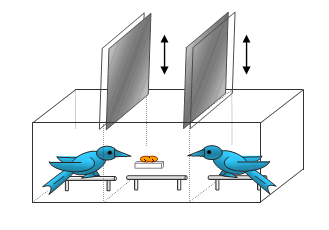
We captured sixteen adult male pinyon jays in northern Arizona and housed them in individual cages in our animal care facility. For this experiment, they were kept mildly hungry by controlling their daily feedings. They were divided into three groups, two of six birds each and one of four birds. Although some of the jays may have known other members of their own group from the wild, the separate groups were generally strangers to one another. To determine dominance, we used a series of staged encounters between pairs of individuals. The two birds were put into a plexiglas chamber with three compartments, separated by opaque and transparent dividers (see illustration below). At the start, each bird was placed by himself in one of the end compartments with all of the dividers lowered. The central compartment was baited with a single peanut (in the shell) that was glued to a shelf to prevent either of the birds from carrying it away. After 10 seconds, the opaque dividers were lifted, so that the birds could see each another and notice the presence of the peanut. After an additional 10 seconds, the transparent dividers were also lifted, giving the birds simultaneous access to the central chamber. Their behavior for the next 5 minutes was recorded on digital videotape. We repeated these encounter sessions six times for each dyad.
We determined the dominance relationships for all pairs within each of the three groups based on the frequencies of five types of behavior during the encounter sessions -- one dominant display (Stare At) and four submissive displays (Look Away, Crouch, Chin-up, and Beg; Marzluff & Balda, 1992; Balda, 2002). Based on a subset of dyads in which the dominance relationship was clear and consistent even at the first encounter, we developed two measures, one of dominance and the other of subordinance, that were weighted sums of the frequencies of the five behaviors. The difference between dominance and subordinance for a given individual was an indication of its relative social status. Based on differences in social status between individuals in all dyads in the sixth and final encounter, we constructed dominance hierarchies for each of the three groups and coded the bird designations accordingly. In all three cases, the hierarchy was linear and fully transitive (Group 1: A>B>C>D>E>F, Group 2: 1>2>3>4>5>6, and Group 3: P>Q>R>S). For 62% of the dyads, stable dominance status was established by the first or second encounter. Where both birds were low ranking, however, the process often took much longer.
Once within-group dominance relationships had been established, we selected eight of the 32 possible cross-group dyads and determined their dominance relationships using the same staged encounter procedure. These provided us with a basis for making predictions about relative dominance across groups. To test the ability to draw social inferences, we designated a set of birds as "observers," and allowed them to watch a series of three-minute encounters between two other birds. For each observer, all encounters involved a specific individual from another group (the "demonstrator"). In each case, the demonstrator was a stranger to the observer, but our cross-group dominance tests indicated that the demonstrator should dominate the observer if they were placed together in the encounter chamber. On each of three consecutive days, observers watched their demonstrator both lose an encounter with one opponent and win an encounter with another. We used this balanced series of winning and losing encounters to make sure that the observer would be making transitive inferences, rather than simply responding to having seen another animal consistently win or lose (Chase et al. 1994; Hogue et al. 1996; Oliveira et al. 1998).
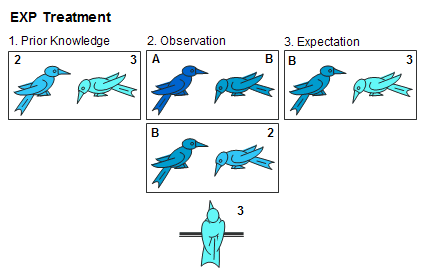
We set up our encounters in a way that systematically controlled the observer’s prior knowledge about one of the demonstrator’s opponents. In experimental (EXP) sessions, the observer had information that would allow him to make inferences from the demonstrator's wins and losses. In control (CTL) sessions, the observer had no prior information about the birds he was watching and could not make meaningful inferences. During EXP sessions, the demonstrator and one of his opponents were strangers to the observer, but the opponent that repeatedly lost to the demonstrator was a familiar bird, one that had repeatedly dominated the observer in earlier staged encounters. For example, suppose that cross-group testing had established that B was dominant to 2. We could then use 3 in an EXP treatment by allowing him to observe A>B and B>2 (see below). In this case, A and B would be strangers to 3, but the relationship 2>3 would have been established in earlier within-group interactions. Using transitive reasoning, 3 should expect to be subordinate to B.
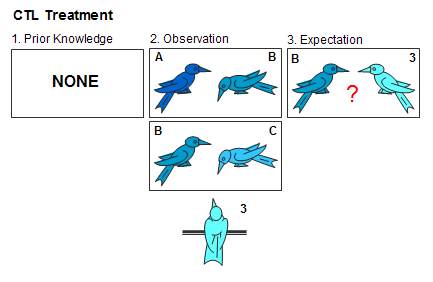
In contrast, during CTL sessions the demonstrator and both of his opponents were all strangers to the observer. If 3 were assigned to the CTL condition, he might watch A>B and B>C, all strangers to him (see below). Because he has no prior knowledge on which to base transitive reasoning, 3 cannot predict his status relative to B.
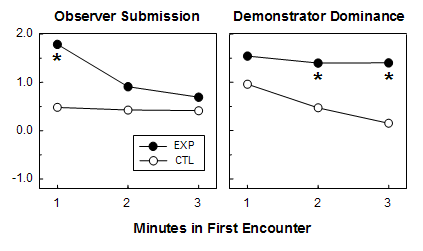
Following the demonstrations, each observer was given test encounters with his demonstrator (testing, in our example, B vs. 3), and the behavior of both participants was evaluated for evidence of transitive social inference. We conducted six sets of EXP and six sets of CTL demonstration and test encounters, each using different observer/demonstrator dyads. To avoid biases, dyads were assigned to treatments in a balanced fashion, so that EXP and CTL dyads had the same average relative social status. If EXP observers use transitive social inference to predict their relationship to the demonstrator, they should, at least initially, show higher levels of submissive behavior than CTL observers. That prediction proved to be true. During the first minute of the first test encounter, EXP observers showed submissive levels that were nearly four times as high as those of CTLs (see below, left panel). Apparently because of the initial submissive displays of EXP observers, EXP demonstrators showed higher levels of dominance than CTLs during the second and third minutes of the first encounter (see below, right panel). The effects on both the observers and demonstrators were transient; there were no significant differences in later encounters between the same birds.
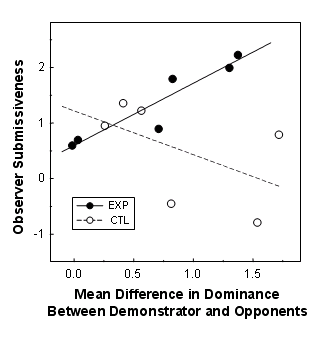
More than just win/loss information was available to the observer birds. The demonstration encounters also displayed the size of the disparity between the demonstrator and his opponents. A strong, aggressive demonstrator might dominate decisively in his winning contests and at least put up a good showing in his losing ones. A weaker demonstrator, in contrast, might barely show superiority in his winning contests and be utterly submissive in his losing ones. Like critical fans at a prize fight, observers could conceivably make more subtle, graded assessments of the demonstrator. They could infer not just the direction of the status difference between themselves and the demonstrator but how large the difference probably was. To see whether observers made use of this information, we calculated the level of dominance behaviors for each demonstrator relative to both of his opponents and determined the degree to which it predicted observer submissiveness during the first test encounter. While there was a strong positive relationship for EXP observers, there was no such relationship for CTL birds (see below). This suggests that observers estimated the actual difference in their dominance status relative to the demonstrator, but only when the losing opponent was known to them.
The results agree with our prediction that pinyon jays should use transitive reasoning to make inferences of relative dominance. Jays that had previously interacted with one of the birds they observed drew inferences about their rank relative to the demonstrator and showed a graded, quantitative response based on their observations. Jays that observed very similar interactions, but had never interacted directly with any of the birds they observed, failed to show either effect. The treatment difference rules out any alternative general explanations, such as badges of status (Rohwer 1982) or simple motivational responses to seeing another bird win or lose (Chase et al. 1994, Hogue et al. 1996, Oliveira et al. 1998). This work constitutes the first demonstration of transitive inference in social settings, and supports the hypothesis that social complexity provided a crucial context for the evolution of cognitive abilities.
References from Other Sources:
Balda, R.P. 2002. Pinyon jay (Gymnorhina cyanocephala). In: The Birds of North America (Ed. by A. Poole & F. Gill), No. 605. Philadelphia, PA: The Birds of North America, Inc.
Chase, I., Bartolomeo, C., & Dugatkin, L.A. 1994. Aggressive interactions and inter-contest interval: how long do winners keep winning? Animal Behaviour, 48, 393-400.
Hogue, M.E., Beaugrand, J.P & Laguë, P.C. 1996. Coherent use of information by hens observing their former dominant defeating or being defeated by a stranger. Behavioural Processes, 38, 241-252.
Kummer, H., Daston, L., Gigerenzer, G. & Silk, J. 1997. The social intelligence hypothesis. In: Human by Nature: Between Biology and the Social Sciences (Ed. by P. Weingart, P. Richerson, S. D. Mitchell & S. Maasen), pp. 157-179. Hillsdale, NJ: Erlbaum.
Marzluff, J.M. & Balda, R.P. 1992. The Pinyon Jay: Behavioral Ecology of a Colonial and Cooperative Corvid. London: T. & A.D. Poyser.
Oliveira, R.F., McGregor, P.K. & Latruffe, C. 1998. Know thine enemy: fighting fish gather information from observing conspecific interactions. Proceedings of the Royal Society of London, B, 265, 1045-1049.
Rohwer, S. 1982. The evolution of reliable and unreliable badges of fighting ability. American Zoologist, 22, 531-546.
Seyfarth, R.M. & Cheney, D.L. 2001. Cognitive strategies and the representation of social relations by monkeys. In: Evolutionary Psychology and Motivation, Nebraska Symposium on Motivation, (Ed. by J. French, A. Kamil & D. Leger), Vol. 47, pp. 145-177. Lincoln, NE: U. of Nebraska Press.
Seyfarth, R. M. & Cheney, D. L. 2003. The structural social knowledge of monkeys. In: Animal Social Complexity (Ed by F.B.M.de Waal & P.L. Tyack), pp. 207-229. Cambridge, MA: Harvard Univ. Press.
de Waal, F.B.M. & Tyack, P.L. 2003. Animal Social Complexity. Cambridge, MA: Harvard Univ. Press.